Abstract
Introduction Over 300,000 infants are born with sickle cell anemia (SCA) in Africa every year, and the under-5 mortality of this population is estimated to be 50-90% without treatment. Hydroxyurea is a safe, oral medication that decreases morbidity and mortality in SCA by increasing protective fetal hemoglobin (HbF) production and reducing inflammation. Hydroxyurea dosing to maximum tolerated dose (MTD) offers the optimal treatment with greatest laboratory responses and clinical benefits. Patients have variable hydroxyurea absorption and elimination rates, so the traditional approach to determining MTD is through incremental dose escalation to achieve mild myelosuppression and reduction in absolute neutrophil count (ANC). In the United States, pharmacokinetic (PK)-guided dosing of hydroxyurea for children with SCA has been shown to decrease time to MTD and to augment treatment responses, with most patients achieving over 30% HbF. Maximizing clinical benefits and minimizing logistical burdens of dosing hydroxyurea would be advantageous for areas with high SCA prevalence, but PK parameters of African children with SCA who might have higher rates of malnutrition, infections, and organ damage that affect drug absorption, distribution, and clearance are yet to be determined. Here we describe hydroxyurea PK exposure-response in Ugandan children with SCA from the Optimizing Hydroxyurea Therapy in Children with Sickle Cell Anemia In Malaria Endemic Areas: The NOHARM MTD Study (NOHARM MTD, NCT03128515).
Methods NOHARM MTD was a randomized controlled trial that compared dosing strategies between hydroxyurea at fixed, weight-based dosing (~20 mg/kg/day) and dose escalation to MTD in children with SCA living in Uganda. NOHARM MTD participants underwent PK testing after 18 months of treatment. Children were administered their prescribed hydroxyurea dose in the morning at clinic, and blood was obtained by fingerstick using Mitra® microsampler devices after 20, 60, and 180 minutes. The microsampler devices were frozen at -80C in Uganda and then transported to Cincinnati Children's Hospital for analysis of hydroxyurea concentration via liquid chromatography with tandem mass spectrometry. Using our previously published population PK model, individual hydroxyurea exposures expressed as area-under-the-curve (AUC) were generated by Bayesian estimation using the PK modeling software NONMEM (Version 7.5, ICON Development Solutions, Ellicott City, USA). A hydroxyurea exposure-response analysis was completed with HbF% as the primary pharmacodynamic outcome.
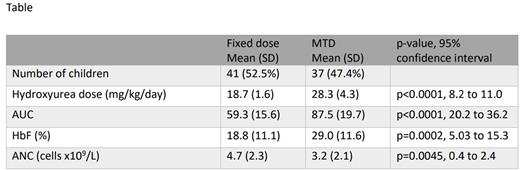
Results A total of 90 NOHARM MTD participants completed PK testing; samples from 12 were excluded from full analysis due to an elevated baseline concentration suggesting the participant already received the daily hydroxyurea dose before PK testing (9), supraphysiologic concentration (1), or mislabeling (2). PK profiles were analyzed for the remaining 78 participants (mean age 6.5 years, SD 0.9); of these, 41 were on fixed dose hydroxyurea and 37 were at MTD with mean doses of 18.7 mg/kg/day (SD 1.6) and 28.3 mg/kg/day (SD 4.3), respectively (Table). Those at MTD had a higher AUC with a significantly higher %HbF (mean 19% HbF on fixed dose vs 29% at MTD, p=0.0002) and lower ANC (mean 4.7 x109/L at fixed dose vs 3.2 at MTD, p=0.0045). Eighteen participants (23%) had malnutrition with a body mass index Z-score less than -1.0, and 12 (15%) participants had renal hyperfiltration with an estimated glomerular filtration rate over 150 mL/min/1.73m2; neither condition was associated with differences in AUC.
Conclusion This study demonstrates the feasibility of collecting hydroxyurea PK data with timed, sparse sampling in Uganda, and the exposure-response analysis confirms that higher hydroxyurea exposure in Ugandan children with SCA is associated with greater induction of protective HbF and reduction in ANC. Malnutrition and glomerular hyperfiltration did not affect hydroxyurea exposure in this cohort. Further studies of PK-guided dosing to optimize response and time to response in this population are needed. Future directions include building capacity for local analysis of hydroxyurea concentrations using HPLC technology, real-time dose calculation via a PK model-informed algorithm, and prospective evaluation of PK-guided dosing.
Disclosures
Ware:BMS: Research Funding; Addmedica: Research Funding; Hemex Health: Research Funding; Nova Laboratories: Membership on an entity's Board of Directors or advisory committees; Novartis: Other: DSMB Chair; Editas: Other: DSMB Chair.
Author notes
Asterisk with author names denotes non-ASH members.